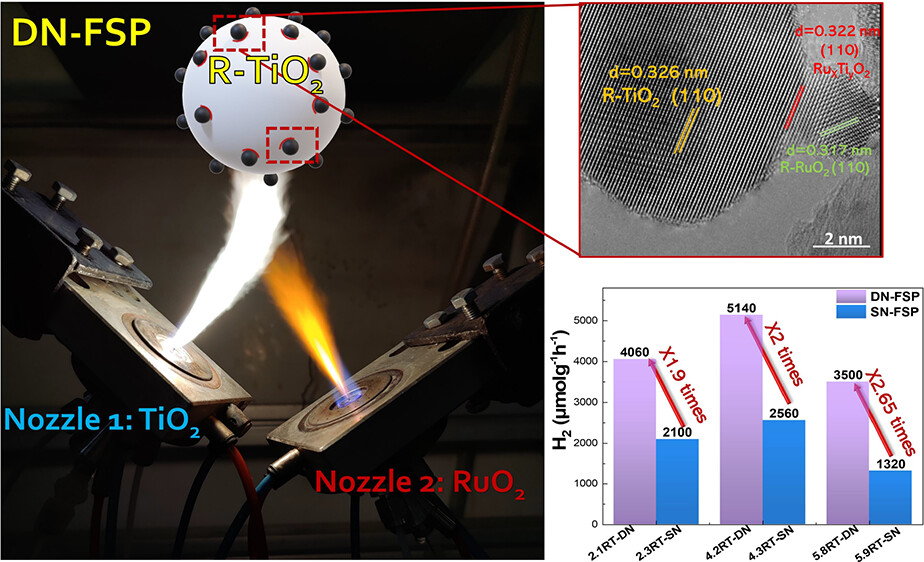
Photocatalytic RuO2/TiO2 nanohybrids have been engineered using flame spray pyrolysis (FSP) technology, enabling the selective formation of either {(rutile)TiO2–(rutile)RuO2} or {(anatase)TiO2–(rutile)RuO2} nanointerphase in a single step. The {(rutile)TiO2–(rutile)RuO2} nanohybrids were selectively produced by using a double-nozzle FSP (DN-FSP) process. The {(anatase)TiO2–(rutile)RuO2} nanohybrids were selectively produced using a single-nozzle FSP (SN-FSP) process. Both RuO2/TiO2 nanohybrids exhibited significant photocatalytic H2 and O2 production from H2O. Notably, the {(rutile)TiO2–(rutile)RuO2} is shown to be a superior photocatalyst, achieving H2 evolution rates exceeding 5144 μmol g–1 h–1 H2 and 2369 μmol g–1 h–1 O2 under solar light illumination. This performance is 50% higher than that of the {(anatase)TiO2–(Rutile)RuO2} configuration. In situ EPR spectroscopy reveals that stable Ti3+/Ru5+ centers are formed during the FSP process, in the {(rutile)TiO2–(rutile)RuO2} interphase. This highlights a unique interfacial electron-transfer event, which is ascribed to a strong oxide (RuO2)/support (TiO2) interaction (SOSI), further validated by transmission electron microscopy/scanning transmission electron microscopy. From a technological perspective, FSP fosters a robust {rutile–rutile} interface between the semimetal–RuO2 and the TiO2 semiconductor, which is beneficial for the separation of interfacial electron–hole pairs onto TiO2 and RuO2 particles. Double-nozzle FSP technology, as demonstrated in this study, offers a promising engineering approach for the industrial production of high-efficiency photocatalytic materials.